DOI:
10.37988/1811-153X_2025_4_142Comparative assessment of the cellular composition of exudate when using different wound dressings after removal of the lower third molar
Downloads
Abstract
Removal of the lower third molar is one of the main methods of surgical treatment of patients with difficult eruption. The problem of postoperative wound management after removal of the lower third molar deserves special attention. When treating a wound under a wound dressing, it is customary to use iodoform turunda, collagen-based dressings. Currently, gelling agents based on calcium alginate are used in surgical practice. A comparative assessment of the cellular composition of the exudate is relevant when using various wound dressings after removal of the third molar of the mandible. The aim of the study was to conduct a comparative evaluation of the results of cytological examination of cellular exudate taken from the wound surface in the postoperative period during surgical dental treatment of patients with difficult eruption of the third lower molar using various wound dressings.Materials and methods.
A study was conducted involving 120 patients who were indicated for complex removal of the lower third molar. The patients were divided into four groups of 30 people, depending on the method of postoperative wound management. Exudate was collected using a sterile latex device to obtain a smear impression on days 1, 3, and 7 after surgery, which was sent for cytological examination. Cytological smears of wound exudate were stained with azur eosin and analyzed with a count of 100 leukocytes.
Results.
Analysis of the inflammatory infiltrate of the socket of the extracted lower third molar showed that when using an iodoform turunda, by the third day there were signs of developing purulent inflammation (erythrocyte aggregation, colonies of microorganisms) and an increasing reaction to a foreign body (increase in the proportion of monocytes) by the third day, while other wound dressings did not cause pronounced signs of a purulent process according to cytological examination data.
Conclusion.
The use of wound dressings based on collagen, calcium alginate, and their combinations showed that the above-mentioned dressings demonstrate comparable results in terms of the level of inflammatory response and cellular composition of exudate, which indicates their equal effectiveness in postoperative wound management and prevention of purulent-inflammatory complications.
Key words:
lower third molar, collagen, iodoform gauze, calcium alginate, wound dressing, cytologyFor Citation
Introduction
Removal of the mandibular third molar is one of the most frequently performed surgical procedures in dental practice [1, 2, 3].
Two principal methods are used for postoperative wound management in the third molar region after complex extraction: closed and open techniques [1, 4—7]. Comparative analysis of these methods indicates that both conditionally hermetic (closed) wound management and open management with tampon placement using various dressings possess specific advantages and limitations. In particular, the closed method is associated with a high incidence of purulent-inflammatory complications reaching 36.8% [1, 2, 3, 6], whereas open wound management with the use of different wound dressings is accompanied by a risk of secondary infection and prolonged healing time [8, 9].
Iodoform turunda and collagen-based wound dressings are most commonly used in surgical dentistry. Despite pronounced antiseptic properties, iodoform turunda exerts a traumatic effect on granulation tissue, leading to its damage, bleeding, and repeated disruption of the blood clot integrity, thereby creating conditions for secondary infection of the wound surface. In addition, its use is associated with pain during dressing changes and prolonged healing periods, which may reach up to 60 days [1, 10, 11]. Collagen dressings, in turn, demonstrate high biocompatibility and stimulate tissue regeneration; however, when applied to “deep” and “contaminated” wounds, including the postoperative socket after complex mandibular third molar extraction [7, 12], they may contribute to the development of purulent-inflammatory complications due to the protein nature of the material and its insufficient mechanical stability [7].
Modern wound dressings based on calcium alginate are of particular interest. Calcium alginate was initially used in surgical practice for hemostasis, temporary wound tamponade, exudate absorption, and protection of tissues adjacent to the wound [13, 15]. Subsequently, wound coverings based on this material were developed and became widely used in the treatment of various types of injuries, including deep wounds involving bone tissue [16]. Calcium alginate dressings exhibit high absorptive capacity, maintain an optimal moist wound environment, and are characterized by pronounced hemostatic and drainage properties [13, 14]. However, data regarding their clinical effectiveness and specific effects on postoperative oral wound tissues, particularly at the cellular level, remain insufficiently studied [15, 16].
Thus, a comparative cytological analysis of the effects of various wound dressings on the wound healing process after complex mandibular third molar extraction appears to be highly relevant.
Materials and methods
The study included 120 patients (47 men and 73 women) aged 18—63 years who were indicated for planned complex extraction of the mandibular third molar. The surgical procedure comprised incision, elevation of a mucoperiosteal flap, osteotomy, tooth sectioning and fragmental removal, curettage of the socket, flap repositioning with suturing, placement of a glove-rubber drain along the vertical incision line, and subsequent postoperative wound management.
Depending on the method of postoperative wound management, patients were randomly assigned to four equal groups of 30 individuals each:
- 12 men and 18 women (mean age 32.5±15 years) — wound management using a calcium alginate-based dressing;
- 11 men and 19 women (mean age 37.5±14.2 years) — wound management using a collagen-based dressing;
- 12 men and 18 women (mean age 39±13.3 years) — wound management using an iodoform turunda;
- 12 men and 18 women (mean age 30±13.8 years) — combined wound management using calcium alginate and collagen-based dressings.
After mandibular third molar extraction, biological material (exudate) was collected on postoperative days 1, 3, and 7 using a sterile latex instrument, followed by preparation of imprint smears of the wound discharge from the surgical site. The glass slides with the applied material were air-dried and submitted for cytological examination.
After drying, cytological smears of wound exudate were stained with azure–eosin. The differential count of formed blood elements was performed by counting 100 leukocytes using a hematological counter.
During statistical data analysis, differences between groups with normal distribution were assessed using Student’s t-test, while nonparametric data were analyzed using the Mann–Whitney U test and Dunn’s method. The level of statistical significance was set at 0.05.
Results and discussion
Group I (calcium alginate wound dressing)
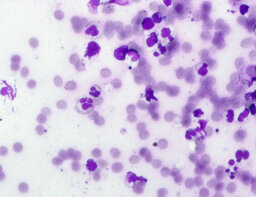
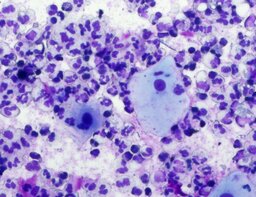
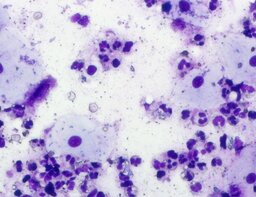
The imprint smear was represented by erythrocytes of the normocyte population (normal discocytes) and leukocytes: neutrophils represented by segmented neutrophils, and agranulocytes represented by lymphocytes and monocytes; eosinophils were not detected in this group. Erythrocytes showed no signs of aggregation; they were normocytic, normochromic, discoid, and without pathological inclusions. The proportion of erythrocytes in the smear was not quantified. Segmented leukocytes exhibited no toxic segmentation and demonstrated normal activity and size. Typical lymphocytes and monocytes of standard size were observed. The preparations contained a moderate number of cells of stratified non-keratinized squamous epithelium (fig. 1).
The distribution of leukocyte population fractions in imprint smears on postoperative days 1, 3, and 7 was comparable. The median proportion of neutrophils was 96.5% on day 1, 97% on day 3, and 96% on day 7. The median proportion of lymphocytes was 2.5% on days 1 and 3 and 3% on day 7. The median proportion of monocytes was 1.5% on day 1 and 1% on days 3 and 7.
A characteristic feature of this group was the relatively uniform distribution of lymphocytes, neutrophils, and monocytes throughout the observation period.
Group II (collagen-based wound dressing)
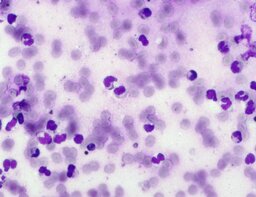
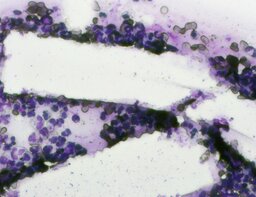
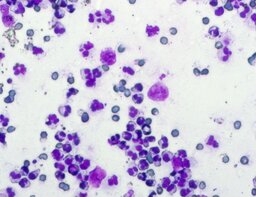
The imprint smear contained normocytic erythrocytes (normal discocytes) and leukocytes: segmented neutrophils, lymphocytes, and monocytes; eosinophils were not detected. Erythrocytes showed no aggregation and had normal morphology without pathological inclusions. The erythrocyte proportion was not determined. Segmented leukocytes showed no toxic changes. Typical lymphocytes and monocytes were present. The preparations contained a moderate number of stratified non-keratinized squamous epithelial cells and, beginning on day 3, isolated rod-shaped bacteria. On day 7, fibers of the dressing material with adherent formed blood elements were observed in some preparations (fig. 2).
The distribution of leukocyte populations on days 1, 3, and 7 was comparable. The median neutrophil proportion was 95.8% on day 1, 93.6% on day 3, and 96.4% on day 7. The median lymphocyte proportion was 2.65% on day 1, 5.2% on day 3, and 3% on day 7. The median monocyte proportion was 1.9% on day 1, 1% on day 3, and 2% on day 7.
A distinguishing feature of this group was relative lymphocytosis and a reduction in neutrophils on day 3. The increased relative lymphocyte proportion may be explained by the decrease in neutrophil proportion
Group III (iodoform turunda)
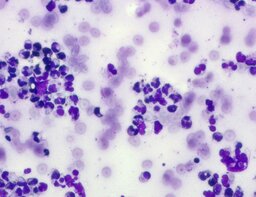
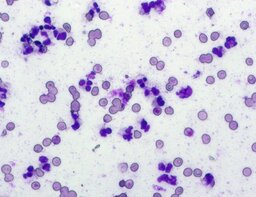
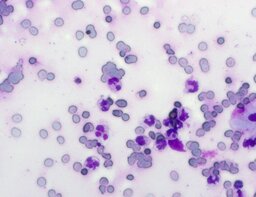
The imprint smear contained normocytic erythrocytes and leukocytes represented by segmented neutrophils, lymphocytes, and monocytes; eosinophils were absent. Erythrocytes showed no aggregation on day 1 but demonstrated aggregation beginning from day 3. Morphology remained normocytic and normochromic without pathological inclusions. The erythrocyte proportion was not quantified. Segmented leukocytes showed no toxic segmentation. Typical lymphocytes and monocytes were present. A moderate number of stratified non-keratinized squamous epithelial cells were observed. By day 3, colonies of rod-shaped and coccal microorganisms appeared in the smears and persisted with predominance by day 7 (fig. 3).
The distribution of leukocyte populations across all observation periods was comparable. The median neutrophil proportion was 93.3% on day 1, 95.5% on day 3, and 94.2% on day 7. The median lymphocyte proportion was 2.5% on days 1 and 3 and 3% on day 7. The median monocyte proportion was 2% on day 1, 1% on day 3, and 2% on day 7.
A characteristic feature of this group was a relative increase in neutrophils on day 3 and relative lymphocytosis with pronounced monocytosis by day 7.
Group IV (combined calcium alginate and collagen wound dressings)
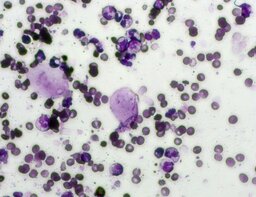
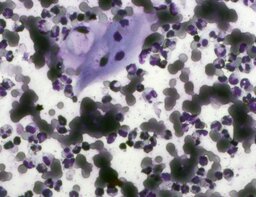
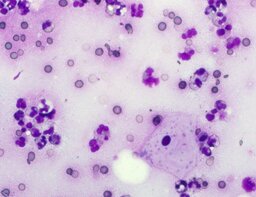
The imprint smear contained normocytic erythrocytes and leukocytes represented by segmented neutrophils, lymphocytes, and monocytes; eosinophils were absent. Erythrocytes showed no aggregation on day 1; mild aggregation was observed by day 3. Morphology remained normal without pathological inclusions. The erythrocyte proportion was not determined. Segmented leukocytes showed normal morphology and activity. Typical lymphocytes and monocytes were observed. A moderate number of stratified non-keratinized squamous epithelial cells were present. By day 7, isolated rod-shaped microorganisms attached to epithelial cells were rarely detected (fig. 4).
The distribution of leukocyte populations across all observation periods was comparable. The median neutrophil proportion was 96.5% on day 1, 98.5% on day 3, and 97.8% on day 7. The median lymphocyte proportion was 1.3% on day 1, 1.4% on day 3, and 1.3% on day 7. The median monocyte proportion was 1% on day 1, 0.5% on day 3, and 1% on day 7.
A characteristic feature of this group was a relative increase in neutrophils on day 3 and relative lymphocytosis by day 7.
Statistical data analysis
The relative proportion of neutrophils from day 1 to day 7 demonstrated several features. On postoperative day 1, the neutrophil count in imprint smears was significantly higher in Group IV, indicating transition to the active inflammatory phase at an early stage of the wound process. On day 3, the collagen-based dressing markedly suppressed inflammation, which manifested as a significant decrease in neutrophil count, whereas in Group IV the activity of the wound process remained stable. On day 7, neutrophil levels in Group IV remained at baseline, similar to Group II. In the iodoform turunda group, the relative neutrophil proportion decreased due to an increase in monocytes, indicating activation of a foreign body inflammatory response (see the table).
Analysis of the relative lymphocyte proportion revealed no statistically significant intergroup differences, as the cytological pattern predominantly corresponded to acute inflammation. Analysis of the relative monocyte proportion demonstrated significantly elevated levels in Groups II and III, most likely reflecting the onset of a foreign body reaction.
| Group I | Group II | Group III | Group IV | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Me | Q1–Q3 | min–max | Me | Q1–Q3 | min–max | Me | Q1–Q3 | min–max | Me | Q1–Q3 | min–max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neutrophils | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 1 | 96.5 | 94–97 | 91–98 | 95.7 | 94.6–96.2 | 94.0–96.8 | 93.3 | 91.2–95.8 | 90.3–97.4 | 98# | 97.4–98.6# | 97.1–99# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 3 | 97 | 95–98 | 92–98 | 93.6# | 92.9–94.4# | 92.1–94.9# | 95.5 | 94.6–96.8 | 94.2–97.8 | 98.5# | 97.3–98.6# | 97.1–99# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 7 | 96 | 95–96 | 93–99 | 96.4* | 95.9–97.2* | 91.0–95.2* | 94.2 | 93.3–95.3 | 92.1–96.7 | 97.8# | 97.2–98.5# | 97.1–99# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymphocytes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 1 | 2.5 | 2–3 | 1–4 | 2.6 | 1.9–3.6 | 1–4 | 2.5 | 1.9–3.2 | 1–4 | 1.3# | 1.2–1.5# | 1–1.9# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 3 | 2.5 | 1–3 | 1–4 | 5.2*# | 4.7–6.5*# | 4.1–7*# | 2.6 | 1.9–3.5 | 1–5 | 1.4# | 1.2–1.5# | 1–1.9# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 7 | 3 | 2–4 | 1–5 | 2.1* | 1.7–2.6* | 1.1–2.8* | 3.2 | 2.7–3.6 | 2.1–3.9 | 1.3 | 1.2–1.8 | 1–1.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monocytes | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 1 | 1.5 | 0–4 | 0–6 | 1.9 | 1.4–2.6 | 1–3 | 2.5* | 1.6–2.5* | 1–9* | 1# | 0–1# | 0–2# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 3 | 1 | 1–2 | 0–4 | 1.5* | 0.5–1.6* | 0.1–2* | 1.5 | 0.6–1.4 | 0.1–2 | 0.5 | 0–1 | 0–2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day 7 | 1 | 1–2 | 0–2 | 2.5* | 1.5–2.5* | 1–2.9* | 2.2* | 1.5–2.8* | 0–3.9* | 1# | 0–1# | 0–2# | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Note: Statistically significant difference (p<0.05): * — compared with the value at the previous time point; # — compared with the value of Group III. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Conclusions
Thus, evaluation of the inflammatory infiltrate in the socket of the extracted mandibular third molar in the presence of different wound dressings indicates that, when an iodoform gauze turunda is used, by day 3 signs of developing purulent inflammation are observed (erythrocyte aggregation, microbial colonies), as well as a progressive foreign-body reaction (increased proportion of monocytes). In contrast, other wound dressings did not demonstrate signs of pronounced purulent inflammation according to cytological findings.
References
- Druzhinin A.E., Lomakin M.V., Soloshchanskij I.I., Kitaev V.A. An improved approach to comprehensive surgical treatment of third molar retention and dystopia. Practical guide. Moscow: Mercury Publishing, 2021. Pp. 4—5 (In Russian).
- Druzhinin A.E., Lomakin M.V., Soloshchanskiĭ I.I., Dunaev M.V., Kitaev V.A., Balukova I.P. The retrospective assessment of the results of the surgical treatment of the patients presenting with retention and dystopia of the lower third molars. Russian Stomatology. 2013; 2: 26—30 (In Russian). eLIBRARY ID: 20810066
- Lomakin M.V., Soloshchanskiĭ I.I., Druzhinin A.E. Control over healing of the bone wound during the surgical treatment of retention and dystopia of the third lower molars. Russian Stomatology. 2014; 2: 4—7 (In Russian). eLIBRARY ID: 22364419
- Sigron G.R., Pourmand P.P., Mache B., Stadlinger B., Locher M.C. The most common complications after wisdom-tooth removal: part 1: a retrospective study of 1,199 cases in the mandible. Swiss Dent J. 2014; 124 (10): 1042—6, 1052—6. PMID: 25342545
- Semkin V.A., Gurin A.N., Vitrenko D.V., Levchenko D.D. Prevention of inflammatory complications after surgical extraction of mandibular third molars. Stomatology. 2022; 3: 38—43 (In Russian). eLIBRARY ID: 48615525
- Velasquez D., Araújo M.G., Clem D.S., Gunsolley J.C., Heard R.H., Janakievski J., McClain P.K., McGuire M.K., Misch C.M., Nevins M., Pickering S., Pope B., Richardson C., Santarelli G., Scheyer E.T., Schallhorn R., Toback G. A Prospective, randomized, controlled, multicenter evaluation of extraction socket management comparing a wound dressing and a soft tissue substitute. Int J Periodontics Restorative Dent. 2022; 42 (6): e161—e174. PMID: 36305922
- Fazal S., Naseer R., Mansoor N., Siddiqi K.M., Sohail K., Hafeez R. Comparison of primary and secondary wound closure outcome after mandibular 3rd molar impaction surgery. Pakistan Oral & Dental Journal. 2021; 41 (3): 169—172.
- Jain S., Kumawat V. Jasleen, Singh A., Rana S.R., Vigneshwar. Comparison of healing of extraction socket with and without suture following third molar surgery: A prospective controlled clinical study. Journal of Research and Advancement in Dentistry. 2017; 6 (3): 35—42.
- Balamurugan R., Zachariah T. Comparison of primary and secondary closure with a buccal mucosal-advancement flap on postoperative course after mandibular impacted third molar surgery. Oral Maxillofac Surg. 2020; 24 (1): 37—43. PMID: 31729607
- Grigoryan A.Yu., Bezhin A.I., Pankrusheva T.A., Sukovatykh B.S. Local management of purulent wounds with wound dressings. Pirogov Russian Journal of Surgery. 2022; 11: 42—48 (In Russian). eLIBRARY ID: 49789737
- Singh V., Das S., Sharma N.K. Iodoform: A boon in disguise. Open Journal of Stomatology. 2012; 2: 322—325. DOI: 10.4236/ojst.2012.24055
- Onyekwelu I., Yakkanti R., Protzer L., Pinkston C.M., Tucker C., Seligson D. Surgical wound classification and surgical site infections in the orthopaedic patient. J Am Acad Orthop Surg Glob Res Rev. 2017; 1 (3): e022. PMID: 30211353
- Hamm R.L. Text and Atlas of Wound Diagnosis and Treatment. McGraw Hill, 2023. Pp. 391—393.
- Chen L., Shen R., Komasa S., Xue Y., Jin B., Hou Y., Okazaki J., Gao J. Drug-loadable calcium alginate hydrogel system for use in oral bone tissue repair. Int J Mol Sci. 2017; 18 (5): 989. PMID: 28481253
- Gustinelli Barbosa M.A., Paggiaro A.O., Fernandes de Carvalho V., Isaac C., Gemperli R. Effects of hydrogel with enriched sodium alginate in wounds of diabetic patients. Plast Surg Nurs. 2020; 40 (2): 110—115. PMID: 32459760
- Budko E.V., Chernikova D.A., Yampolsky L.M., Yatsyuk V.Y. Local hemostatic agents and ways of their improvement. I.P. Pavlov Russian Medical Biological Herald. 2019; 2: 274—285 (In Russian). eLIBRARY ID: 38220877